One YMC

YMC America recently hosted colleagues from Japan for a two-week training on YMC’s patented process scale Contichrom TWIN MCSGP technology to support the growing demand in Asia. We may live on different continents, but we are one YMC family!
YMC strives to provide high quality products and support to all of our customers around the world. The training completed by our colleagues will ensure that every system like the one shown below can be supported with YMC care and expertise. This custom-designed Contichrom TWIN 500 MCSGP HPLC recently left our production facility in Devens, MA, USA, headed for its new home!
Related Posts

April 23, 2026
Eliminating False Positives in PFOS Analysis: The Power of Methanol-Free Separation
Accurate determination of 𝗽𝗲𝗿𝗳𝗹𝘂𝗼𝗿𝗼𝗼𝗰𝘁𝗮𝗻𝗲𝘀𝘂𝗹𝗳𝗼𝗻𝗶𝗰 𝗮𝗰𝗶𝗱 (𝗣𝗙𝗢𝗦) in food samples is challenging due to bile acid–related compounds (e.g., taurocholic acid isomers). These species can produce the same SRM transition (m/z 499→80) as PFOS, leading to potential false positives without adequate chromatographic separation.

April 21, 2026
Using Volunteer Day to help Build Affordable Housing
We are incredibly proud to share how Victor Mansfield recently used his corporate Volunteer Day to make a life-changing difference in the Dominican Republic.
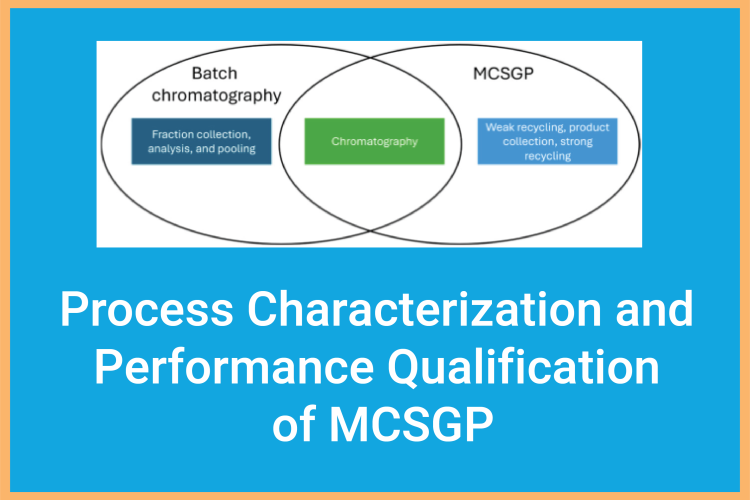
March 24, 2026
Revolutionizing Peptide Purification: A Blueprint for MCSGP Process Validation
The shift from batch to continuous manufacturing is a requirement in many biopharma applications, but regulatory compliance, specifically Process Validation, often remains a hurdle.


