Revolutionizing Peptide Purification: A Blueprint for MCSGP Process Validation
Revolutionizing Peptide Purification: A Blueprint for MCSGP Process Validation
The shift from batch to continuous manufacturing is a requirement in many biopharma applications, but regulatory compliance, specifically Process Validation, remains a hurdle for many.
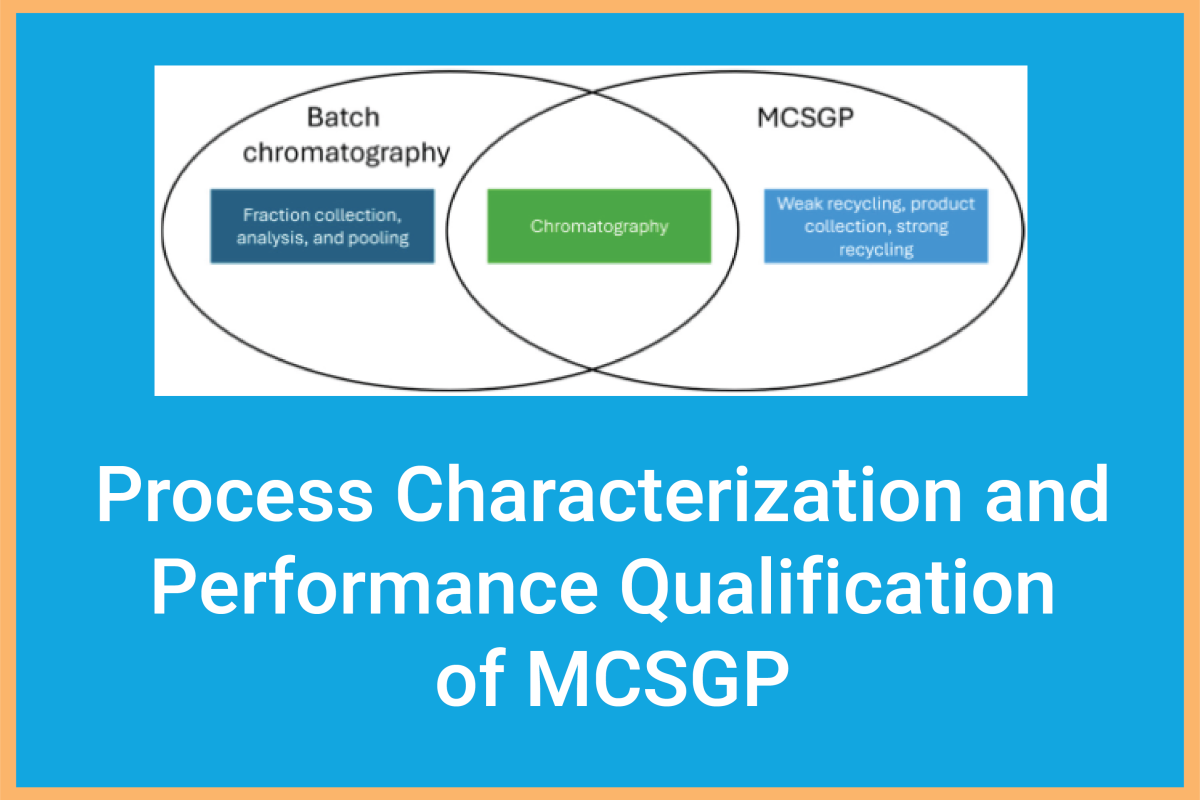
A recent milestone study by Ralf Eisenhuth (Bachem AG) and Thomas Müller-Späth (YMC ChromaCon) provides the first-ever comprehensive framework for the Process Characterization (PC) and Process Performance Qualification (PPQ) of MCSGP (Multicolumn Countercurrent Solvent Gradient Purification).
Using the synthetic peptide Bivalirudin as a test case, the authors demonstrate how to transition this continuous technology into a fully regulated, commercial-scale environment.
Highlights of this article include:
- Comprehensive Framework for PC and PPQ of MCSGP
- Defining Criticality: Out of 34 identified process parameters, only the gradient slope was classified as a Critical Process Parameter (CPP), requiring tighter control limits.
- Process Analytical Technology (PAT) in Action: The study highlights AutoPeak® technology as a critical PAT. It uses real-time UV triggers to adjust for peak shifts, ensuring consistent internal recycling and product collection without manual intervention.
- Efficiency Gains: Compared to traditional batch chromatography with extensive side-cut recycling, the MCSGP process achieved:
- Yield Increase: From 57% to 62% (gross-to-gross).
- Process Mass Intensity Reduction: PMI dropped from ~5200 to 1400 kg/kg—a massive win for sustainability.
- Simplified QC: In-process controls (IPCs) were slashed from 81 per kg to just 3.2 per kg.
- Robust Control: Supported by online measurements, MCSGP is capable of running uninterrupted over several days.
Why This Matters:
This work provides, for the first time, a stringent pathway to form the foundation of the Process Validation lifecycle of therapeutics produced via continuous chromatography. It proves that MCSGP with AutoPeak® technology isn’t just a lab-scale efficiency tool; it is a robust, validated, and highly sustainable solution for commercial peptide and oligonucleotide manufacturing.
Related Posts
May 21, 2026
Breaking Down mRNA Capping Efficiency with High-Resolution IP-RP LC

May 7, 2026
Revolutionizing CRISPR Applications: High-Resolution sgRNA Analysis

April 23, 2026
